The Role of Laboratory Testing in Breast Cancer Diagnosis and Management 
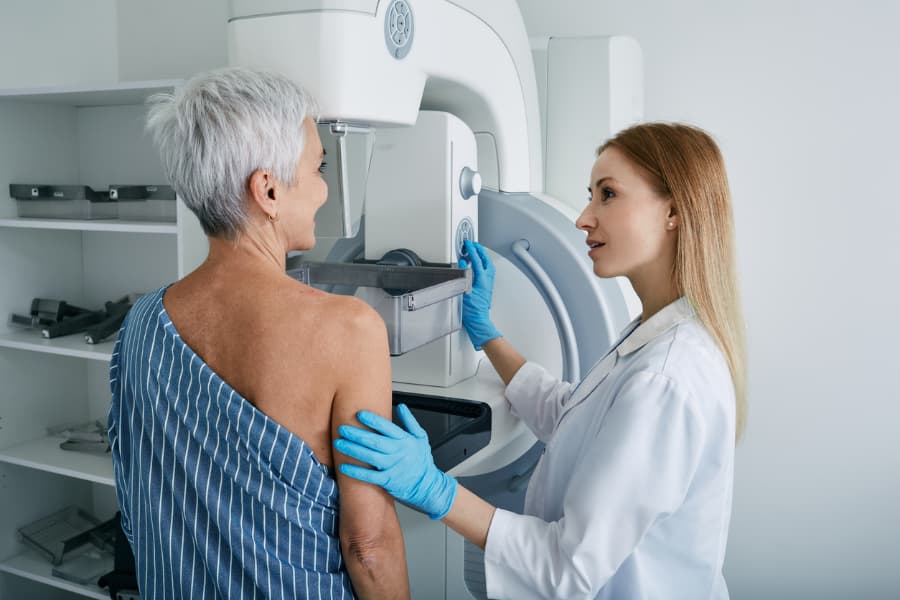
Breast cancer remains a leading cause of morbidity and mortality worldwide. In the U.S., one in eight women is expected to develop breast cancer in their lifetime. Laboratory testing is indispensable in the diagnosis and management of this disease. In recognition of National Breast Cancer Awareness Month, observed each October, this blog highlights the key…